Tuesday, November 16, 2010
NEXT ECO-FASCIST SCARE STORY DIES
Google records 530,000 hits for ocean acidification with, predictably, the BBC in 3rd place saying it is one of the century's "biggest environmental concerns".
The theory being spread by the fear industry is that the increase (of 1 part in 10,000) of atmospheric CO2 might increase absorption in sea water so that it becomes marginally more acidic (actually sea water is alkaline so that would mean becoming marginally more neutral) which might, possibly, have so effect on sea life, which would be bound to be negative with the result that we are all DOOMED.
It has similarities to, though much more dilute than, the acid rain scare which proved to be wholly untrue. It turned out to be beneficial, something all the media, politicians & indeed Wikipedia then decided not to mention.
Here is Rational Optimist giving the facts:
As opinion polls reveal that global warming is losing traction on the public imagination, environmental pressure groups have been cranking the engine on this `other carbon dioxide problem’...
The trouble is, a shoal of new scientific papers points to the conclusion that this scare is based on faulty biochemical reasoning, unrealistic experiments and exaggeration...
the European Project on Ocean Acidification (EPOCA) is now a consortium of over 100 scientists from 27 institutes and 9 countries. This last summer it funded 35 scientists to spend six weeks in the Arctic studying the problem, `assisted’ by Greenpeace’s ship Esperanza. Think how little incentive the scientists would have to say `sorry, lads, we realize it is a not much of an issue after all’.
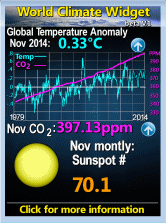
Start with a few facts. The oceans are not acid but alkaline, with an average pH of about 8.15 (0-7 being acid, 7-14 being alkaline). But they vary both in space and time, Arctic seas being less strongly alkaline than tropical, and some bays and reefs being actually acid because of underwater volcanic emissions. The dissolution of carbon dioxide in the oceans may lower the pH slightly to about 7.9 or 7.8 by the end of the century at the worst – still alkaline.
Environmentalists like to call this a 30% increase in acidity, because it sounds more scary than a 0.3 point (out of 14) decrease in alkalinity, but no matter. It is still well within the bounds of normal variation over space and time: the pH of the water intake at the Monterey aquarium varies by almost twice as much as this every month. The difference between the pH of the seas off Hawaii and Alaska is greater than this.
Enough numbers. Try chemistry. The scary reasoning rests on the argument that lower pH will mean less dissolved carbonate in the water. But a new paper from scientists in North Carolina proves what many scientists have long suspected, namely that corals and other species do not use carbonate as raw material to make their shells; they use bicarbonate. And dissolving carbon dioxide in water actually increases bicarbonate concentrations.
This may explain why study after study keeps finding that far from depressing growth rates of marine organisms, high but realistic levels of carbon dioxide either do not affect them or increase them...
Before I started looking into this, I assumed the evidence for damage from ocean acidification must be strong because that is what the media kept saying. I am amazed by what I have found. Make no mistake: there are lots of threats to the ecosystems of the ocean, from over-fishing to nutrient run-off, but acidification is way down the list. The attention is deflecting funds and action from greater threats. It is time scientists had the courage to admit this.
The theory being spread by the fear industry is that the increase (of 1 part in 10,000) of atmospheric CO2 might increase absorption in sea water so that it becomes marginally more acidic (actually sea water is alkaline so that would mean becoming marginally more neutral) which might, possibly, have so effect on sea life, which would be bound to be negative with the result that we are all DOOMED.
It has similarities to, though much more dilute than, the acid rain scare which proved to be wholly untrue. It turned out to be beneficial, something all the media, politicians & indeed Wikipedia then decided not to mention.
Here is Rational Optimist giving the facts:
As opinion polls reveal that global warming is losing traction on the public imagination, environmental pressure groups have been cranking the engine on this `other carbon dioxide problem’...
The trouble is, a shoal of new scientific papers points to the conclusion that this scare is based on faulty biochemical reasoning, unrealistic experiments and exaggeration...
the European Project on Ocean Acidification (EPOCA) is now a consortium of over 100 scientists from 27 institutes and 9 countries. This last summer it funded 35 scientists to spend six weeks in the Arctic studying the problem, `assisted’ by Greenpeace’s ship Esperanza. Think how little incentive the scientists would have to say `sorry, lads, we realize it is a not much of an issue after all’.
Start with a few facts. The oceans are not acid but alkaline, with an average pH of about 8.15 (0-7 being acid, 7-14 being alkaline). But they vary both in space and time, Arctic seas being less strongly alkaline than tropical, and some bays and reefs being actually acid because of underwater volcanic emissions. The dissolution of carbon dioxide in the oceans may lower the pH slightly to about 7.9 or 7.8 by the end of the century at the worst – still alkaline.
Environmentalists like to call this a 30% increase in acidity, because it sounds more scary than a 0.3 point (out of 14) decrease in alkalinity, but no matter. It is still well within the bounds of normal variation over space and time: the pH of the water intake at the Monterey aquarium varies by almost twice as much as this every month. The difference between the pH of the seas off Hawaii and Alaska is greater than this.
Enough numbers. Try chemistry. The scary reasoning rests on the argument that lower pH will mean less dissolved carbonate in the water. But a new paper from scientists in North Carolina proves what many scientists have long suspected, namely that corals and other species do not use carbonate as raw material to make their shells; they use bicarbonate. And dissolving carbon dioxide in water actually increases bicarbonate concentrations.
This may explain why study after study keeps finding that far from depressing growth rates of marine organisms, high but realistic levels of carbon dioxide either do not affect them or increase them...
Before I started looking into this, I assumed the evidence for damage from ocean acidification must be strong because that is what the media kept saying. I am amazed by what I have found. Make no mistake: there are lots of threats to the ecosystems of the ocean, from over-fishing to nutrient run-off, but acidification is way down the list. The attention is deflecting funds and action from greater threats. It is time scientists had the courage to admit this.
Labels: eco-fascism, Media, Science/technology
Comments:
<< Home
Over fishing is the big threat simply because, no-one officially owns (and therefors manages) the oceans. Fishermen have the shoplifters incentive to grab all they can ASAP. Wow, collective ownership being part of the problem, who would have thought it?
Incidentally, if there ever was a serious problem, who would take this lot seriously anymore having cried wolf do many times.
Incidentally, if there ever was a serious problem, who would take this lot seriously anymore having cried wolf do many times.
At least half the fish sold are now farmed ones. Agriculture being more efficient than nomadic hunting - a revolutionary idea.
I think there are real threats - a Carrington Event solar flare, a new infectious disease transferred across the world in hours, wars caused by demographic growth or a chunk of comet debris. These don't tick the alarmist's boxes. In any case their solution to every problem is medievalism plus more government regulation which is always the wrong solution.
Post a Comment
I think there are real threats - a Carrington Event solar flare, a new infectious disease transferred across the world in hours, wars caused by demographic growth or a chunk of comet debris. These don't tick the alarmist's boxes. In any case their solution to every problem is medievalism plus more government regulation which is always the wrong solution.
<< Home

